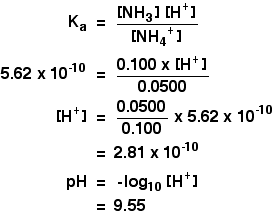Pufferfish Rh glycoprotein-mediated uptake of the ammonia analog, [ 14... | Download Scientific Diagram

Multiple alignment and hydropathy plot of pufferfish Rh glycoproteins.... | Download Scientific Diagram
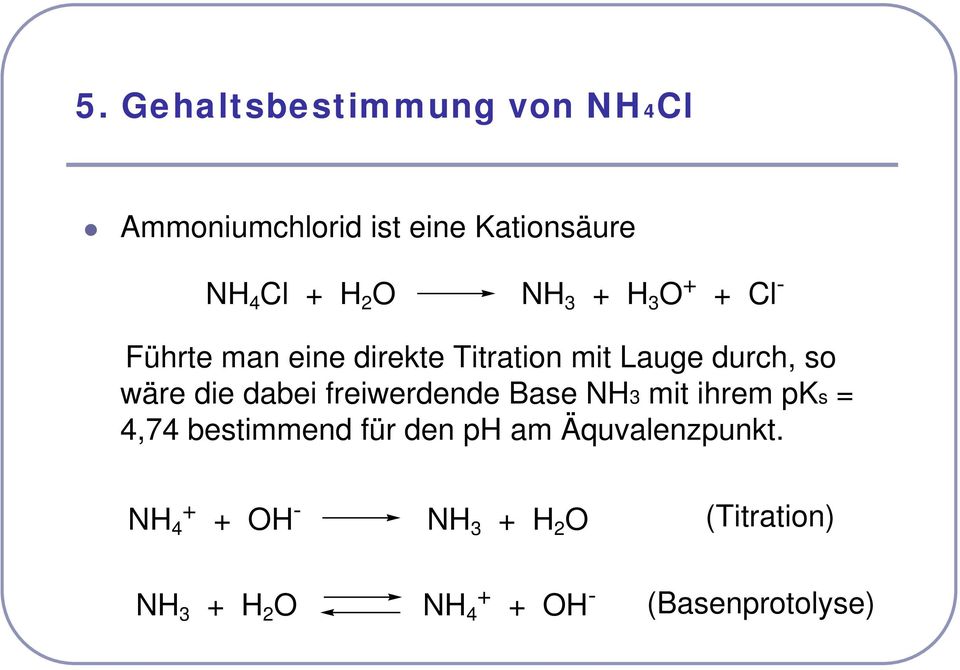
Säure-Base Titrationen. (Seminar zu den Übungen zur quantitativen Bestimmung von Arznei-, Hilfs- und Schadstoffen) - PDF Free Download

Multiple alignment and hydropathy plot of pufferfish Rh glycoproteins.... | Download Scientific Diagram

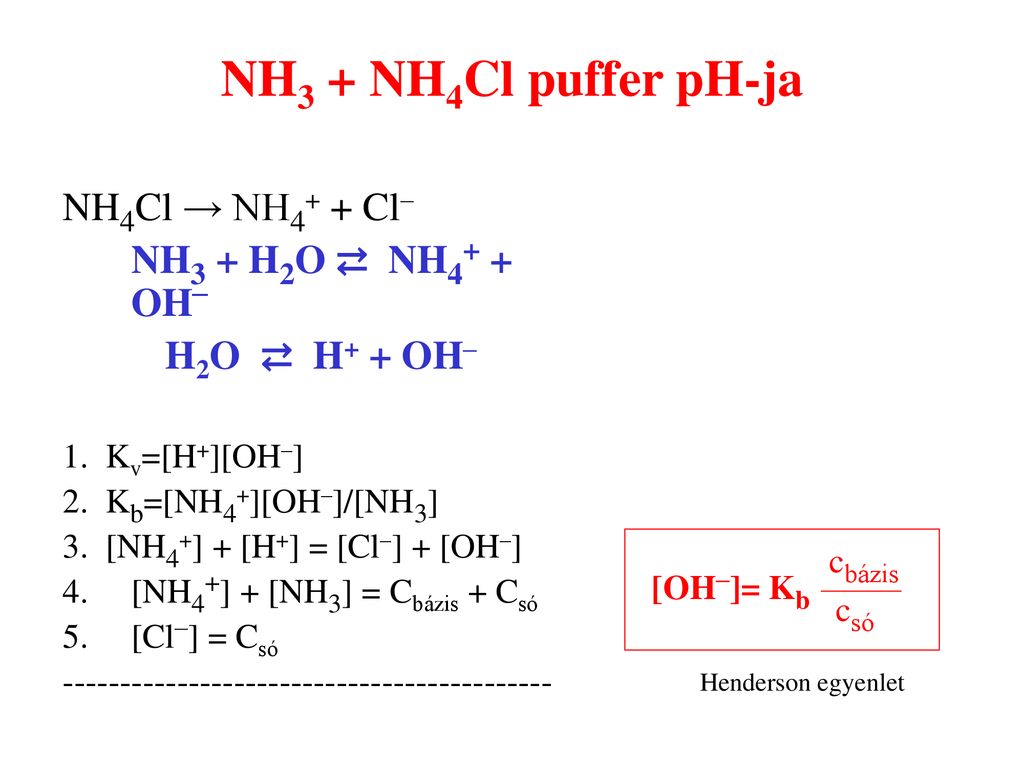
SOLVED:24. What is the change in pH when 40.0 mg of NaOH is added to 100 mL of a buffer solution consisting of 0.165 M NH3 and 0.120 M NH4Cl? a) Which

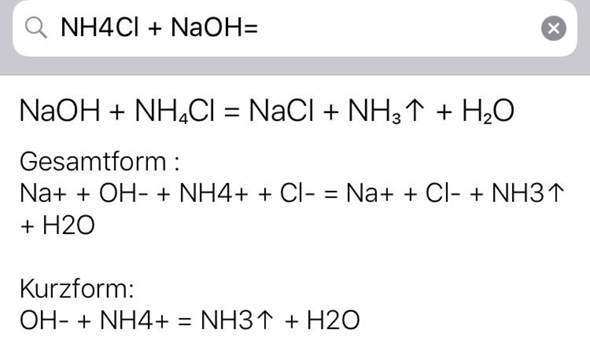
SOLVED:In step 4 of the qualitative analysis, if you incorrectly used NaOH to raise the pH to 8-9 (instead of using an NHAOH NHACIl buffer as instructed), which ion would you probably

Solve the following systems of equations using elimination method. 0.5x + 0.8y = 0.44, 0.8x + 0.6y = 0.5

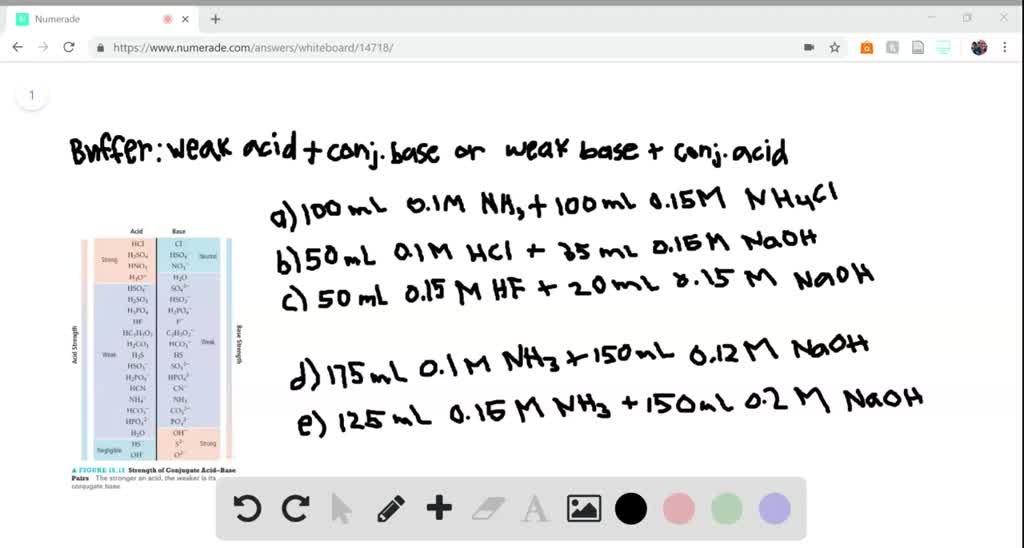
SOLVED:Determine whether the mixing of each pair of solutions results in a buffer. a. 100.0 mL of 0.10 M NH3; 100.0 mL of 0.15 M NH4Cl b. 50.0 mL of 0.10 M